Shortly after Alexander Lin, MD, joined UCSF Benioff Children’s Hospitals in 2022, a 12-year-old boy was flown in for emergency surgery. The child had been run over by a vehicle, and nearly every bone in his face was shattered.
As co-director of UC San Francisco’s Craniofacial Center, Lin was the lead plastic surgeon on the case. He had his work cut out for him. The child had suffered severe fractures to his eye sockets, cheeks, nose, and jaws. His eyebrows and lips were torn apart. His forehead bone – which surgeons elsewhere had removed when his brain began to swell – would need to be reconstructed entirely. He’d also lost vision in his left eye, and ophthalmologists suspected that one of his optic nerves had been injured. The vision loss could be permanent.
“It was a difficult, difficult case,” Lin says. “Reconstructing this child’s face bones would be like fixing a broken egg that’s covered by a blanket. I can’t just cut open his skin to see the fractures and fix them. I have to make little hidden incisions under his eyelids, mouth, and lips. I’m basically making holes in the blanket and then trying to fix the egg through the holes.”

The mystery of the optic nerve made the case even more complex. Computerized tomography (CT) scans didn’t show exactly what was crushing the nerve. Without that knowledge, any attempt to repair it could cause further, possibly irreparable, damage.
Lin needed to see more. He printed a three-dimensional (3D) model of the boy’s skull – a physical replica constructed from the patient’s CT scans that would allow him to examine the eye socket by hand. Deep within the orbital cavity, he found the culprit: A small piece of broken bone had jammed the optic nerve.
“It was so far back,” Lin recalls. “There were all these tiny pieces of bone, much further back than most eye socket fractures. But when I held the model, I saw that I could reach down with my finger and move this one piece of bone away from the nerve. I don’t know how I could have visualized that without 3D printing.”
Hours later, Lin was in the OR. A student held up the 3D model nearby so Lin could refer to it during the procedure. “It was like having X-ray vision,” he says. On the model, the “blanket” covering the boy’s injuries was gone and the cracks in the “egg” were in plain sight, with color-coded landmarks guiding his every move. One by one, Lin repaired the multiple fractures.
The optic nerve was next. Peter Sun, MD, the Chen Professor of Clinical Neurosurgery, stepped in to assist. Sun gently lifted the child’s brain, and Lin lowered his finger into the back of the eye socket. Even amid so much bone and blood, he was able to see his target. His muscle memory guiding him, Lin placed his finger on the fragment of bone, rotated it away from the nerve, and attached it to the skull.
Eight weeks later, the patient’s vision returned to his left eye. “With a result that extraordinary,” Lin says, “it’s hard to imagine how we could have done it any other way.”
Lin explains how a 3D-printed model of his patient's skull helped guide the surgery that saved the child’s vision. Video: Walter Zarnowitz; Tom Seawell | Explore the virtual skull model

UCSF’s 3D printers live in a small, cluttered lab on the Mission Bay campus. The large, black machines sit side by side amid stacks of boxes, tools, and other equipment.
On a recent day, one of the machines was whirring steadily. A printhead the size of a car battery moved back and forth above a pale, yellow shape. From left to right, it deposited a thin layer of liquid plastic atop the object; from right to left, it shone green ultraviolet light over the liquid, hardening it, before depositing the next layer. Over the next 10 hours, hundreds of these layers came together to form a 3D model of 12-year-old Samantha Rodriquez’s heart.
Three-dimensional printing transforms computerized images into tangible objects. “It’s like raised topographical maps of the 19th century,” says orthopaedic surgeon Alan Dang, MD ’06. “Layers of material are stacked on each other, some forming islands, some mountains. When it’s done, you have a three-dimensional map.”
In medicine, 3D modeling can merge hundreds of individual slices from a scan such as a CT or magnetic resonance imaging (MRI) to form a three-dimensional replica of anything from a vital organ to a skeletal structure to a cardiovascular network. The resulting model can be explored virtually on a computer screen or be printed and examined physically.
“This technology literally lets me put the anatomy into the surgeon’s hand,” says Shafkat Anwar, MD, medical director and co-founder of UCSF’s Center for Advanced 3D+ Technologies (CA3D+). “It lets us take a newer, deeper look at what’s happening and make much more sophisticated decisions about what to do next.”

Established in 2018, CA3D+ represents a unique collaboration between surgeons, imaging experts, and engineers – applying advanced 3D visualization and manufacturing techniques to solve some of the most complex clinical cases at UCSF. In the past six years, CA3D+ has produced more than 700 3D models and become a national leader in using the techniques in medicine.
Before Anwar co-founded CA3D+, the technology was still relatively new in medicine. It was just over a decade ago that specialists in radiology, like Jesse Courtier, MD, started harnessing the power of three-dimensional augmented reality in medical modeling, and Alan Dang and his brother, Alexis Dang, MD ’04, also an orthopaedic surgeon, created UCSF’s first anatomical 3D print.

The Dang brothers had always loved to tinker with computers – they built motherboards in middle school and programmed computers in college. So as 3D printing began to take off in the popular interest, their curiosity was piqued: How might they use 3D technology in orthopaedics?
“There was all this hype,” Alan Dang says. “Even Martha Stewart was developing a line of 3D-printed products. So I bought the cheapest 3D printer I could find. We didn’t even know what we could do with it. We just thought, if anybody can make this work, it’s us.”
With no user manual for how to replicate broken bones, the brothers applied trial and error and scoured message boards for tips. “We ended up learning a lot from hobbyists, including those in the ‘maker’ and right-to-repair movements,” he says, referring to recent technology-based extensions of the do-it-yourself ethos.
Their first model was of a broken clavicle. It took a few attempts to get the print right – the computer kept trying to correct the fracture as an error in the data – but when the object finally took shape, the brothers were immediately struck by its potential.
“With a CT scan, your brain is integrating about 300 images to create that model in your mind,” says Alexis Dang. “With 3D printing, you can hold the model in your hand. Rotate it. Get a sense of scale, of where you need to be – a thumb or a finger’s breadth away from some anatomical landmark. You’re preparing your muscle memory for the actual surgery.”


The CA3D+ office is across campus from the printer lab. Anwar shares the space with Michael Bunker, the center’s 3D printing engineer. Bunker has not only spent years building medical devices from scratch but he’s also a seasoned gamer, a hobby that gives him a strong feel for 3D work.
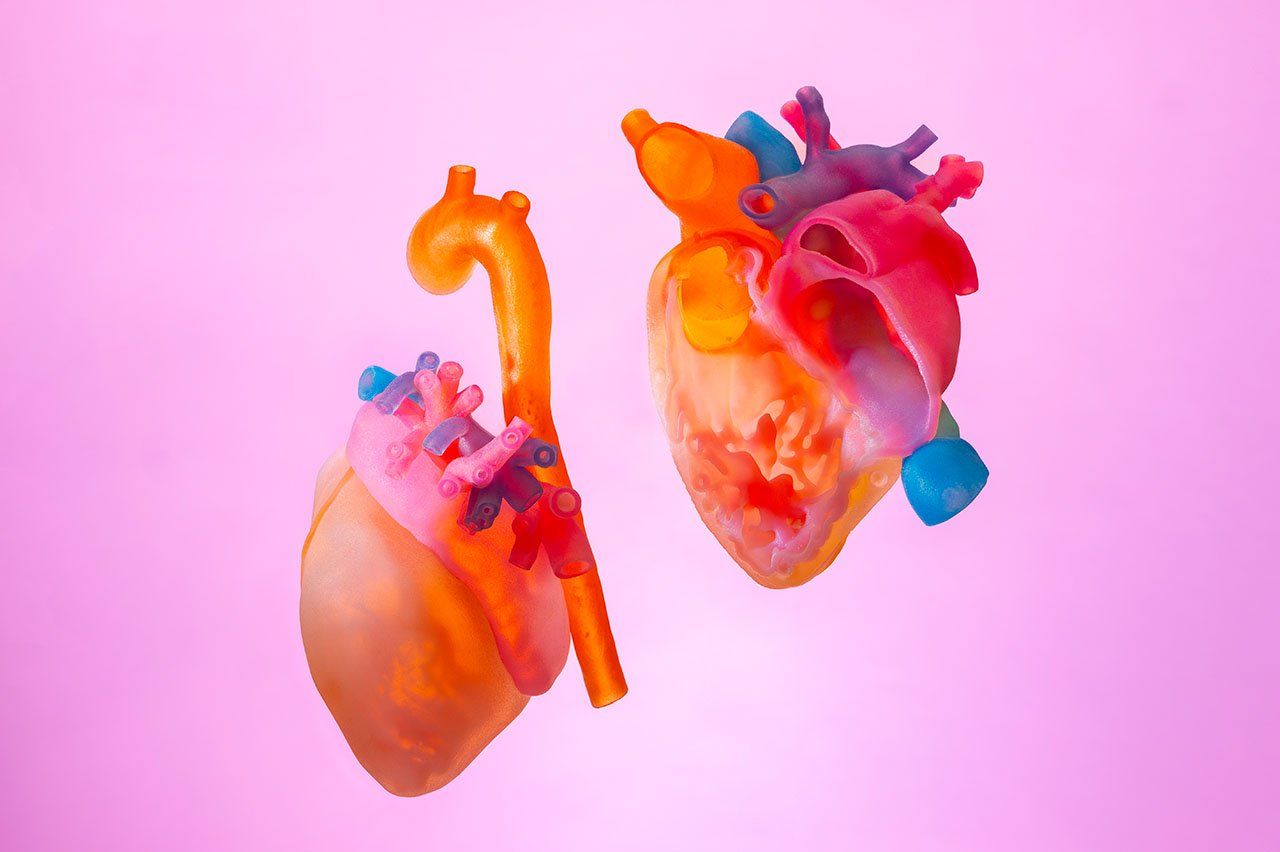
In between Anwar’s and Bunker’s desks is the center’s “library”: an astounding array of colorful organs, bones, muscles, and molecules. There are walnut-sized infant hearts revealing complex structural defects; knotty tumors buried deep within labyrinths of vital organs; and curvy spinal cords joined by teal, fuchsia, pink, and purple vertebrae.
Drawing on his expertise as a pediatric cardiologist and cardiac imaging specialist, Anwar works closely with Bunker to design models that reduce the mystery of interpretation for surgeons. The models transform unique anatomy into detailed, manipulable road maps indicating the path a surgeon should take inside the body and revealing which biological landmarks show the way.
Anwar scans the library and pulls out a tangle of blood vessels resembling a deciduous tree in winter, with colored branches giving the structure a psychedelic effect. “This one isolates out the blood vessels so they’re easier to see,” he says. “We color-coded the vessels and told the surgeon, ‘This is what you need to go after,’” he says as he touches the blue and pink branches. “‘Collect these vessels and bring them here. You’ll find them in front of this landmark.’” He points at a knob jutting off a yellow branch.
In other cases, a model can reveal the feasibility of a risky procedure. That was the case for Samantha Rodriquez, who was born with single-ventricle heart disease, a rare defect that left her with only half of her heart to pump. By the time she was 6, Samantha had already endured three open heart surgeries. In the years that followed, her health stabilized, but her long-term prognosis remained precarious. Pediatric heart surgeon Mohan Reddy, MD, who specializes in repairing congenital cardiac defects, had to decide whether to perform a fourth, extremely delicate and high-risk procedure to cure the disease – or do nothing and hope for the best.

Using data from high-resolution 3D imaging, Anwar and Bunker printed a model that put Samantha’s malformed heart into sharp relief. Reddy took the heart in his hand, opened it up, and explored its inner workings. He could see a clear way to construct a two-ventricle repair, a pathway that had remained elusive on CT scans prior to the creation of the 3D model.
The surgery was a success, and Samantha is feeling better than ever. In September, Anwar presented her heart model at the World Congress of Pediatric Cardiology and Cardiothoracic Surgery to illustrate the enormous potential of 3D technology in heart medicine. At UCSF, surgeons now use 3D modeling to evaluate nearly every child who is born with complex congenital heart disease and faces high-risk surgery.
Anwar, who is also a cardiac imaging specialist, explains how a 3D heart model helped surgeon Mohan Reddy find a way to perform an extremely delicate procedure to repair a young girl’s heart. Video: Walter Zarnowitz; Tom Seawell | Explore the virtual heart model

Over the years, the Dang brothers have become convinced that 3D printing isn’t just for complex medical problems. They believe that every surgery – no matter how unremarkable the case – can benefit from 3D modeling.
Take recurrent shoulder dislocation, an injury that Alexis Dang, a specialist in sports medicine, sees often. Doctors determine the appropriate treatment – whether a nonoperative intervention like physical therapy, an arthroscopic surgical procedure, or more invasive open surgery – based on the amount of bone loss at the edge of the glenoid, the socket portion of the shoulder joint.
For years, orthopaedic surgeons relied on CT scans to assess bone loss. “But the problem with CT scans is that they just give you an image of a slice of anatomy,” says Alexis Dang. “When you’re imaging these slices, if you’re slightly off in your orientation, it can skew your measuring. When it comes to bone loss, the amount of loss might appear differently based on how you cut the slice.”
He lines up three glenoid prints on his desk, each about the size of a fist. The models conjure up his brother’s analogy of raised topographic maps: the layers of plastic forming the joint look like sedimentary rock.
“You can see the variation between these models,” Alexis Dang says, pointing to where the humerus meets the shoulder socket. “And you can feel it. When you move the humerus, you can tell how stable it is or how easily it falls out of place. Being able to feel it and touch it makes a big difference. These prints tell me how and when to treat the patient.”
The Dangs hope to continue demonstrating the versatility and breadth of 3D modeling as a surgical planning tool. “There are so many applications out there that we never would have thought of,” Alexis Dang says. “At the beginning of the pandemic, we used 3D printers to address supply chain issues by printing face shields and replacement parts for our air purifiers. We never would have thought to do this if not for the unique circumstances.
“Our goal is to get as many people as possible from across the UCSF community – not just clinicians but also researchers and students, and beyond medicine, in dentistry and nursing – to use the models so we can discover even more applications for our patients.”

For Anwar, too, surgical planning is just the tip of the iceberg.
Back at the pediatric craniofacial center, a few months after regaining his eyesight, Lin’s 12-year-old patient underwent another surgery to replace his missing forehead and eye socket, which were destroyed in the accident. This time, Lin used sterile guides 3D printed in an external laboratory to cut precise pieces of bone from uninjured parts of the child’s skull to create a new forehead and eye socket.
What makes these prints unique is that they were sterilized for safe use in the patient’s body during surgery. In the near future, Anwar and Lin hope to bring this sterilization process in-house. This will include making sterilizable 3D parts in UCSF’s current facilities and adding a new facility at UCSF Benioff Children’s Hospital Oakland for building these tools.

Anwar even has his sights set on eventually moving into four-dimensional anatomical modeling. “Our hearts and our bodies are moving through time and space,” he explains. “We want to build models that show us how our bodies perform when they’re in motion – that’s the fourth dimension.”
He takes a heart model in hand. “This is the heart when it’s still, but what will it look like when it’s contracting? How will blood flow through it? If we can integrate that physiology into our modeling, we can learn so much more. We can predict the outcome of one surgery compared to another.”
For now, Anwar is excited to nurture the exponential growth of 3D printing in medicine, and the groundbreaking subspecialty that he, the Dang brothers, Lin, Courtier, Bunker, and others have built at UCSF.
“Ten years ago, I never could have imagined that we would be here,” he says. “We’ve gone from talking about cardiac 3D printing as a cool new tool to showing that it’s possible, across multiple disciplines, to support an entire health system and hundreds of patients with this technology.”
Meanwhile, back in the lab, the 3D printers are churning out another one of Anwar and Bunker’s models. They will use this one to discern if a tiny neonate’s circulation can be rerouted to correct a complex malformation. Anwar can’t wait to get his hands on it and start visualizing the surgery.