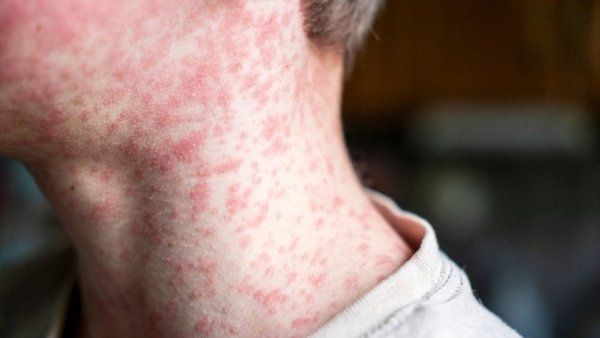
Five-year-old Elianna loves running around with her big brother in their yard on the Hawaiian island of Kauai. She isn’t afraid to get dirty, is learning to ride a bike, and has her doting grandfather wrapped around her little finger. It’s a delightfully ordinary childhood – but one that almost didn’t exist.

When Elianna’s mom, Nichelle Obar, was pregnant with her, an ultrasound revealed that her tiny heart was enlarged and fluid surrounded her lungs. Follow-up testing confirmed a shattering diagnosis: alpha thalassemia major. The inherited disorder disrupts the production of hemoglobin, the protein in red blood cells that’s responsible for delivering oxygen throughout the body. Without treatment, most fetuses with the condition die before birth.
“I was devastated, thinking that my child would not survive,” Obar recalls. “But whether it was perfect timing or divine intervention, our genetic counselor happened to be attending a webinar that week with Dr. MacKenzie.”
Tippi MacKenzie, MD, a pediatric and fetal surgeon, directs the UCSF Center for Maternal-Fetal Precision Medicine and the Eli and Edythe Broad Center of Regeneration Medicine and Stem Cell Research. In 2017, when Obar first heard about her, MacKenzie was just beginning a clinical trial to evaluate a pioneering way of treating alpha thalassemia in the womb. Experts have known for decades that in utero blood transfusions, delivered through a vein in the umbilical cord, can save a sick fetus’s life. But children born with the condition still need treatment throughout their lives, and the procedure isn’t offered everywhere; many expectant women never even learn it’s an option until it’s too late.
MacKenzie’s trial was offering transfusions, along with something new: a stem cell transplant that could potentially cure the disease. After getting in touch with the UC San Francisco team, Obar and her husband, Chris Constantino, pored over the trial details. They learned that doctors would insert a needle into the center of Obar’s hip bone to collect her bone marrow, the spongy tissue that produces stem cells. The doctors would then process the marrow to extract these immature cells and inject them into her umbilical vein. If all went according to plan, Obar’s stem cells would move into her fetus’s bone marrow, where they would mature into blood cells – including the healthy red cells the baby needed.
The couple also learned that the trial was based on extensive research that MacKenzie and her colleagues had done in animals. “The first thing I asked was, ‘Have there been any human trials?’” Obar recalls.
“No,” MacKenzie replied. “You’d be the first.”
Have there been any human trials?”
Obar asked.
“No,” MacKenzie replied.
“You’d be the first.”
The response gave Obar pause. But MacKenzie, the John G. Bowes Distinguished Professor and a Benioff UCSF Professor, doesn’t shy away from firsts. More than four decades ago, her role model, Michael Harrison, MD, performed the world’s first successful fetal surgery at UCSF. MacKenzie is now continuing that legacy, looking beyond performing operations in the womb to delivering medical procedures that treat genetic diseases before a child is born. She envisions a future where fetal therapy is routine for correcting inherited disorders, including rare diseases like Elianna’s and more familiar ones like cystic fibrosis and sickle cell disease.
Tippi MacKenzie and Elianna’s parents, Nichelle Obar and Chris Constantino, reflect on the journey that gave Elianna a chance at life. Video: Corduroy Media & Walter Zarnowitz
When MacKenzie talks about her research, her dark eyes light up behind her round glasses. But when asked to describe what makes her successful, her gaze falters. She has no interest in claiming center stage. “This isn’t my work,” she stresses. “There’s an enormous team of people that makes this possible.”
Her colleagues, on the other hand, have no trouble shining the spotlight on her. “Tippi is a brilliant scientist, and her work is truly cutting-edge,” says her collaborator Juan Gonzalez-Velez, MD, PhD. A UCSF maternal-fetal medicine specialist, he performed the in utero blood transfusion and stem cell transplant for Elianna and has continued to do so for subsequent babies in MacKenzie’s studies. “It opens up a whole new avenue for prenatal treatments, which could be expanded to so many other disorders.”
Fetal Surgery is Born
In 1981, after testing fetal surgery in pregnant sheep, Harrison was granted institutional approval to operate on a human fetus. The first, historic procedure – to reroute a blocked urinary tract – saved a boy’s life. Harrison went on to found the UCSF Fetal Treatment Center at Benioff Children’s Hospitals, where he and his colleagues set the bar for operating on the tiniest, most fragile patients. During one of those cases, as Harrison carefully sliced a tumor from an unborn baby, an eager medical student named Tippi watched from the corner of the OR, transfixed.
Born Tippi Cicek, she spent her early childhood in Istanbul, Turkey, before her family moved to the U.S. when she was 11. She was a voracious learner: “Reading, math, science – I loved all of it,” she says. Also a talented classical pianist, she studied at the Julliard School of Music. But fate had another plan for her capable hands.
After college at Harvard, where she majored in biochemistry, she moved west for medical school at Stanford. There, she heard a surgical resident utter the phrase “fetal surgery,” which stopped her in her tracks. “Oh, my God, people do that?” she asked him. When she learned that Harrison had pioneered the field, she picked up the phone and arranged to do a month-long rotation at UCSF. “Watching Dr. Harrison resect a tumor from a fetus was the most extraordinary thing,” she says. “I remember being in that room and thinking, ‘This is what I want to do.’”
Around the same time, in 1996, she read an article in the New England Journal of Medicine describing the first in utero stem cell transplant. Alan Flake, MD, a pediatric and fetal surgeon at Children’s Hospital of Philadelphia (CHOP), and his colleagues had performed the procedure on a fetus with severe combined immunodeficiency (SCID). Children with SCID are born with virtually no immune system. Defenseless to infection, few survive their first year. By transplanting stem cells taken from the bone marrow of the child’s father, Flake’s team appeared to have found a cure.
That paper stayed in the back of MacKenzie’s mind as she continued her training. During her surgical residency at Harvard’s Brigham and Women’s Hospital, she reached out to Flake to ask about research opportunities. “It was before email, so I sent him a fax,” she laughs. That led to a three-year fellowship studying stem cell transplantation and gene therapy with Flake at CHOP, where she also received additional training in pediatric and fetal surgery.
Her time in Flake’s lab started MacKenzie on a line of research she’s continued to this day. But as she and others in the field would soon discover, fetal stem cell treatments turned out to be much more complicated than that first test case had suggested.
Of Mice and Mothers
After Flake’s success curing a fetus of SCID, researchers tried transplanting stem cells into unborn babies with a variety of blood and other immune disorders. “Most of the time, it just didn’t work,” MacKenzie says. “And that was a bit of a mystery.”
But the fact that it had worked for SCID – a disease that wipes out the immune system – offered an important clue. The human immune system is built to be wary, attacking anything it identifies as foreign. Before a child or adult undergoes a stem cell transplant, they must take powerful drugs to prevent their immune system from waging war on the donor cells. When MacKenzie began her research, most experts believed that the fetal immune system was immature and therefore wouldn’t reject stem cells from a donor. But SCID aside, that’s exactly what seemed to be happening.
Finally, in 2008, UCSF infectious disease specialist Mike McCune, MD, PhD, now retired from the University, made a surprising discovery. During pregnancy, some of the mother’s cells pass into her developing child through the placenta. The fetus exists in harmony with these cells, McCune showed, by manufactuing special immune cells whose job is to dial down its own immune response. “That’s part of why pregnancies can be successful even though mother and child are not genetically identical,” MacKenzie says. “We’ve always intuitively understood it’s important that the mom doesn’t reject the fetus, but it’s just as important for the fetus to not reject the mom.”
For years, most physicians attempting fetal stem cell transplants had taken the cells from someone other than the mother. “Nobody wanted to harvest cells from a pregnant woman,” MacKenzie says. “But if we know the fetus doesn’t reject Mom,” she wondered at the time, “why aren’t we using maternal cells?” This, she suspected, could be a more reliable approach.
She tested the theory in her lab, and it was soon clear she was on the right path. Transplants in fetal mice were more successful with maternal cells than with nonmaternal ones, which were destroyed soon after being transplanted. But MacKenzie also found something else remarkable: The immune cells of the fetal mice weren’t the only ones killing off the nonmaternal cells. The mother’s immune cells, wandering freely inside the fetus, also swooped in to join the assault – another reason to transplant maternal cells.
Those experiments were the basis for the clinical trial that ended up saving Elianna’s life. Obar was about halfway through her pregnancy when she learned about MacKenzie’s study, and her unborn baby was growing sicker by the day. She and Constantino packed their bags and flew to California.
Elianna was the first of six fetuses treated in the UCSF trial. All the babies were born at full term and are doing well, according to MacKenzie, who continues to monitor their progress. The study was a phase I trial, so its main goal was to assess whether in utero stem cell transplants are safe; the team is now evaluating their effectiveness.

Fetal therapy may have given these children a chance at life, but it’s not yet the cure that MacKenzie is aiming for. Five years later, Elianna still needs blood transfusions every month or so. Not enough of Obar’s cells survived in her daughter to heal her completely.
In retrospect, MacKenzie says, alpha thalassemia may have been a tricky test case to start with. The decision had been a practical one. Some physicians, like Gonzalez-Velez, were already treating the condition with fetal blood transfusions, so adding stem cells into the mix would likely pose little additional safety risk. But, as is often true in medical science, the biology of the disease turned out to be thornier than MacKenzie had anticipated.
Because alpha thalassemia deprives a fetus of oxygen, the unborn baby’s bone marrow seems to churn out extra red blood cells in an attempt to overcome the deficit. The proliferation of native red cells might prevent donor cells from establishing themselves, MacKenzie reasons. “These fetuses are so sick, and their own bone marrow is so revved up, that it may be hard for the maternal cells to survive,” she says.
Even so, her trial has helped raise awareness that alpha thalassemia is no longer a hopeless diagnosis. Despite the availability of fetal blood transfusions, many doctors still believe the disease is too severe to treat, MacKenzie says. For reasons that baffle her, some pregnant women are never even told treatment is an option, and their fetuses die in the womb. “It’s heartbreaking how these patients have been ignored by the medical community, even though with blood transfusions they can survive to birth and live as normal kids,” she says. “The most rewarding thing has been shining a light on this condition.”
MacKenzie is now preparing the trial results for publication, and she’s hopeful they will lay the groundwork to move ahead with trials of fetal stem cell treatments for other inherited diseases. She’s eyeing other forms of thalassemia, as well as Fanconi anemia, a rare and life-threatening disorder that affects all types of blood cells. “Even if these diseases are rare individually, they add up to a lot of conditions that we could treat before birth,” she says.
Even if these diseases are rare individually, they add up to a lot of conditions that we could treat before birth.”
Tippi MacKenzie
Into the Unknown
One thing that’s certain about focusing on fetal therapy: You have to be willing to venture into the unknown. Harrison waded in when he made the case for operating on a fetus with no other options. Years later, he recognized the same quality in MacKenzie and helped recruit her to UCSF. “I always thought Tippi had tremendous potential,” he says. “Something about her said she’d take on the hard challenges. She just does it, and she doesn’t blink.”
Only three years after meeting Elianna’s family, MacKenzie launched another groundbreaking clinical trial. This one would be the first in the world to test fetal therapy for a group of inherited metabolic disorders called lysosomal storage diseases (LSDs). People with LSDs don’t make enzymes that break down waste in cells. As a result, toxins build up in their tissues, causing symptoms that can include organ damage, skeletal abnormalities, muscle weakness, intellectual disabilities, and developmental delays. The effects are debilitating and often fatal.
Synthetic enzyme therapies are available for a handful of LSDs. Given regularly through intravenous infusions, these treatments can slow a disease’s progression. But even when therapy is started shortly after birth, irreversible damage may already have been done. MacKenzie had shown it was safe to inject stem cells into fetuses. Why not do the same with enzymes?
Once again, she took the question into the lab. As she had hoped, fetal enzyme therapy helped prevent organ damage in mice with LSDs. And there was another benefit to the early intervention. Many LSDs cause neurological problems, and most enzyme therapies given after birth can’t cross into the brain. During pregnancy, however, a fetus’s blood-brain barrier hasn’t yet formed. MacKenzie showed that enzymes given to mice in utero could migrate to their brain cells and clear out any toxic buildup there.
Based on those results, the U.S. Food and Drug Administration approved MacKenzie’s human trial in 2020. Getting permission from the FDA to test a fetal therapy is an enormous challenge – even more so in this case because MacKenzie’s team wanted to test the therapy not just in one disease but in the eight LSDs for which enzyme therapies are available. “Tippi is probably the most effective person I’ve ever worked with,” says Paul Harmatz, MD, a UCSF gastroenterologist who specializes in LSDs and is collaborating with her on the project. “I’m still amazed we were able to get this trial approved.”
MacKenzie’s unwavering determination may be the reason a Canadian toddler is alive today. The child’s parents had lost two daughters to an LSD known as severe infantile Pompe disease when they learned their unborn baby had inherited the same metabolic disorder. This time, though, there was reason for hope.
But just as the trial was scheduled to begin, the COVID-19 pandemic threw a curveball into the team’s plans. With borders locked down, the Canadian family couldn’t travel to California. Undeterred, MacKenzie shared the details of her treatment protocol with the family’s Canadian medical team and helped them gain regulatory approval to give the therapy off-label, outside the UCSF trial. That baby is now a toddler who babbles and walks and smiles – milestones that few babies with severe infantile Pompe disease ever reach.
A Leap of Faith
Since the Canadian case, MacKenzie and her colleagues have treated two patients with LSDs as part of the UCSF trial, and they plan to enroll eight more. Even if they prove that fetal enzyme therapy can slow disease progression and prevent organ damage, it won’t be a cure. After the children are born, they will continue to need regular enzyme replacements.
But it’s an exciting time for LSD treatment, Harmatz says. Several promising therapies that can cross the blood-brain barrier are now in clinical trials or likely to be approved soon. And with new treatments poised to improve long-term outcomes for many children with LSDs, fetal therapies may one day play an even bigger role in preventing serious symptoms before birth. “This can be a bridge to a cure,” Harmatz says. “It’s a huge step forward.”

Some of the next advances may come from MacKenzie’s lab. “There’s a lot coming down the pike,” she says. Among the many lines of research she’s exploring is gene therapy, which involves editing or replacing gene variants that cause diseases like alpha thalassemia. She’s also testing fetal infusions of RNA-based drugs in mice. These drugs could potentially treat inherited neurological diseases by switching genes on and off or blocking the production of harmful proteins. And she continues to study how maternal and fetal cells coexist – and whether a breakdown in the immune cease-fire may play a role in kickstarting preterm labor.
It’s a lot to juggle, especially for a mom of two busy teenage daughters. To fit it all in, she made the hard decision in 2021 to set down her scalpel and give up operating. “Being a surgeon is central to my identity,” she says, though it was never her singular focus. Her patients have always been her North Star.
Because the fetuses in her clinical trials have genetic conditions, many of the families she cares for have already lost one or more pregnancies or have other children or relatives who are affected. MacKenzie’s work requires hard conversations – and decisions that are harder still. Her job, she says, is to help families decide what’s right for them, whether that’s trying an experimental fetal therapy, waiting until after birth to begin treatment, or terminating the pregnancy. “The central tenet of fetal therapy,” she emphasizes, “is to present all the options and let the pregnant woman choose.”
After all, MacKenzie says, she’s not the star of this story. “To have that leap of faith…” she says. “I think of my patients as the heroes.”