Scientists used to believe that our neurologic fate was sealed at birth with a single, lifetime allotment of brain cells.
The thinking went – not so very long ago – that little by little, with the bumps of age and lifestyle, this initial stash of neurons died, taking our brain function along with them. Yet, strange as it may sound, canaries, video games, and young blood are finally putting that punishing prospect to rest. Studies involving bird song, gaming, and the rejuvenating factors of young blood have shown not only that neurons can be generated throughout adulthood, but also that the maddening aspects of aging, such as memory loss and slower processing speed, can be partially reversed.

Arturo Alvarez-Buylla, PhD. Photo: Cindy Chew
Both neuroscientists and coal miners revere the canary, but for entirely different reasons. Like humans, canaries are known in neuro-science as “open learners,” meaning they learn throughout adulthood. “Canaries learn songs, like we learn language, from older adults when young,” explains Arturo Alvarez-Buylla, PhD, a stem cell neurobiologist at UC San Francisco. As they get older, they tweak their songs seasonally to distinguish themselves during mating season. Alvarez-Buylla’s mentor, neuroscientist Fernando Nottebohm, PhD, a professor at Rockefeller University, suspected that as these parts of the canary brain assembled and disassembled for the yearly acquisition of the new mating song, new neurons were being taken on board. Such a notion was unimaginable at the time.
“When Nottebohm proved by morphology, electrophysiology, electron micro-scopy, and connectivity between 1983 and 1986 that the new cells were neurons, the whole field of stem cell science became a lot more exciting,” recalls Alvarez-Buylla, who holds the Heather and Melanie Muss Endowed Chair in the Department of Neurological Surgery. The jaws of neuroscientists throughout the world dropped at the possibilities posed by Nottebohm’s finding. Neurogenesis offered a new way to repair damage wrought by age, neurological injury, or disease. Alvarez-Buylla went on to advance the field ever further by identifying the neural stem cell, its origins, and its behavior in the mammalian brain.
Blood Shot
Now, decades later, young blood – literally speaking – has joined the canary as a harbinger and waypoint en route to realizing the promise of neurologic rejuvenation.
Last spring, UCSF Faculty Fellow Saul Villeda, PhD, published a study in Nature Medicine showing significant signs of reversal of age-related cognitive decline in old mice after they were infused with the blood of young mice over the course of several weeks. Two other studies showing the revitalizing effects of young blood in brain and muscle tissue were published at the same time.

Saul Villeda, PhD. Photo: Elisabeth Fall
“All three studies coming out simultaneously made things go supernova,” says Villeda, who, at the age of 33, is a bit of a young blood himself. In the media frenzy that followed, Villeda was inundated with requests for interviews, in both English and Spanish. Born and raised in East Los Angeles, Villeda was able to deliver in both languages. “What we were saying collectively, across three impressive institutions – UCSF, Stanford, and Harvard – is that there is reversibility in the aging process. It’s a bit of a game changer.”
The experiment itself proved quite easy for Villeda to explain to the lay press. He and the graduate students in his lab took the blood of young mice, stripped it of its cells, and infused the remaining plasma into old mice. They did this every three days for 24 days, using small injections of the plasma each time – just 5 percent of a mouse’s blood volume. The young mice in the study were 3 months old, the equivalent of humans in their 20s, and the old mice were 18 months old, the equivalent of humans in their 60s.
Days later, he tested them for cognitive changes. In one experiment, the mice had to wind through a water maze and remember where a dry platform was hidden; in another, the mice had to recall a location where they had received a shock. “When we gave them the injections of young blood, they no longer had the cognitive impairments of a normally aging mouse,” says Villeda. “Their performance wasn’t quite equal to the young mice, but pretty close.”
The two experiments tested the functioning of the hippocampus, a part of the brain, in both mice and humans, that is especially affected by normal aging. It’s our hippocampus that we use in searching for our car in a crowded parking lot. When we park, our brain, without prompting, will note spatial cues in the environment and keep them in mind to guide us back to the same place hours later. But the older we are, the more likely we are to forget those spatial cues, throw in the towel, and press the panic button to find the car.
“As we get older, we have fewer stem cells and newly born neurons in our brains, and our learning and memory are affected,” says Villeda. “It’s not dementia, it’s just the natural degeneration associated with age.”
Flipping Switches
Clearly, the young blood helped turn back the clock for Villeda’s old mice. So he began searching for molecular and biochemical changes in their brains that might explain the transformation. To accomplish this, he used the somewhat macabre technique of parabiosis, which involves sewing a young mouse to an old mouse so they share a single blood supply. After a month, he sequenced the genes of the old mice and found that the biggest changes occurred in genes associated with neuronal plasticity, the brain’s response to learning. When we are learning or responding to our environment, our brain either increases the number of connections among neurons or strengthens our existing neuronal connections. “Normally, with aging, the activity of genes that control synaptic plasticity decreases,” says Villeda. “We saw that exposure to young blood increased the expression or activity of these genes.”
The old mice with the new high-octane blood were blazing through the mazes because their neurons were making new connections, and solidifying previous connections, with the vigor of mice less than half their age. Villeda and his students searched the gene array for some sort of mechanism that might be responsible for the surge of neuroplasticity in these middle-aged mice who, without the blood infusion, might still be trapped in the maze. The patterns of activated genes and changes they found looked to Villeda like the work of a master regulator known as CREB.
“CREB is an old friend of neuroscience,” he explains. “We know that it’s very important for learning and memory, especially during development.” To figure out the extent of CREB’s role, a student in Villeda’s lab manufactured a virus carrying a phosphate that would turn CREB off, then repeated the blood-infusion experiments on mice lacking this master regulator. In the new experiments, the old mice with young blood gained some benefits of youth, but the effect was significantly dampened. The experiments showed clearly that CREB is important – but that it doesn’t work alone.
“Now we know that as we get older, we are not necessarily losing the genes or proteins in our brains that we need to improve cognition. Maybe, like CREB, they are just not as active,” says Villeda. “We’ve identified one part of the mechanism to wake up the brain. Now we have to find the other genes it works with to replicate the full effect.”
Hold or Reset?
Villeda is quite excited at the prospect of applying these findings to humans – a sentiment surely shared by anyone over the age of 40. “We know rejuvenation exists,” he says. “Now we have to figure out the bare minimum of therapeutics or genetic tinkering necessary for it to be safely translated into a human. There are so many questions we have yet to answer.”
We know rejuvenation exists. Now we have to figure out the bare minimum of therapeutics or genetic tinkering necessary for it to be safely translated into a human.”
For example: What part of plasma is really driving the changes, and are they lasting? Mice only live an average of 3 years; we live 80. How often would humans have to be treated, and when should treatments start? Cell proliferation slows in old age, perhaps to offset cancer risk. Would young blood factors stimulate cancer? If so, it might be more prudent to switch off the mechanisms that initiate the aging cascade.
“People who have a genetic predisposition for Alzheimer’s have a mutation, but they don’t get the effects until later in life, which means that something in their young bodies knew how to fight it or compensate for it,” says Villeda. “If we could reverse some of the aging signs, perhaps we could maintain ourselves at a younger stage and then maybe not have to deal with diseases until far later in life.”
Game On
While Villeda is turning back the clock in his cohort of mice, Adam Gazzaley, MD, PhD, is beating back cognitive decline with a joystick. Dressed in a black shirt and sleek gray blazer, Gazzaley looks more like a biotech executive than a neuroscientist. It turns out he’s both. Gazzaley rocked the world of neuroscience last fall with the release of a video game, NeuroRacer, that dramatically improved cognitive performance in elderly players. In the game, players drive a car along a winding track, while various signs flash into view along the way. Players are instructed to press a button when a specific sign pops up, ignoring the rest, all while keeping their eyes on the road.
“We developed NeuroRacer to put pressure on cognitive control abilities in a powerful way in older adults, who we know have deficits in this domain just by virtue of their age,” says Gazzaley. “The results were better than we even dreamed of.” After one month and just 12 hours of training, players who were between 60 and 85 years old were scoring as well as 20-somethings who had just learned the game. And, retested six months later, the players were still holding onto those gains.

Adam Gazzaley, MD, PhD. Photo: Susan Merrell
The cognitive skills Gazzaley aimed to improve with his game are selective attention, sustained attention, working memory, and task switching. “We are building a tool to help people develop the cognitive control skills they need to interact with their environment based on their goals,” says Gazzaley. “If we’re trying to do too many things at once and can’t hold our attention to something we want to focus on, then all aspects of our lives suffer, whether it is family, work, safety, or even entertainment.”
Using EEGs, Gazzaley was able to show increased brain activity in the prefrontal cortex of the older players. After they played the game, their EEGs started to resemble those of 20-somethings. The prefrontal cortex, considered the seat of cognitive control, is the last part of our brains to develop – at around age 25. It is also the area that distinguishes humans from all other species.
The EEGs showed signs of connectivity to other parts of the brain as well. Like Villeda’s mice and Nottebohm’s canaries, Gazzaley’s game-players were enhancing their neuroplasticity, adding new connections while strengthening existing ones. He confirmed these gains by testing other areas of cognition. When assigned a facial recognition challenge, Gazzaley’s players showed improvements in working memory. This showed that the benefits of game play were transferable to other brain functions. Transfer, considered the gold standard for effectiveness in the field, is evidence of underlying neural connections among different areas of cognition. “That’s exactly what we wanted to achieve – to see if we could change the brain in a meaningful way,” says Gazzaley, “and have that accompanied by changes in cognitive abilities that we weren’t directly targeting.”
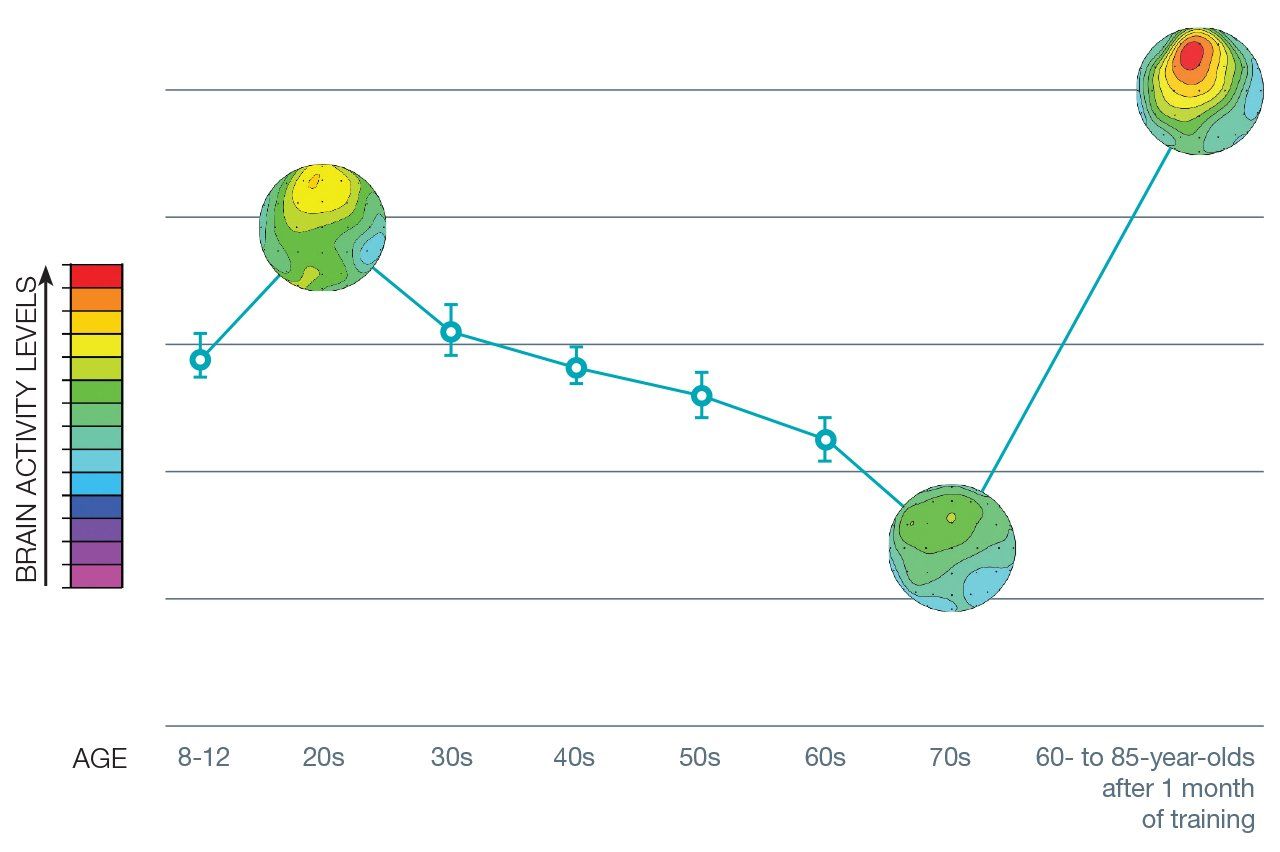
80 IS THE NEW 20: The brain’s command center for multitasking is in the prefrontal cortex. The brain scan on the left depicts the prefrontal cortex activity of Gazzaley’s 20-year-old subjects as they played NeuroRacer, a video game that involves multitasking. The scan in the middle depicts the starting point for his 60- to 85-year-old players, playing the game the first time. And the scan on the right shows the progress the older players made after playing a total of just 12 hours over the course of a month. Their scans showed signatures of brain activity comparable to that of the 20-year-olds who had played the game once.
NeuroRacer is clearly not your ordinary video game, in which users try to reach ever-higher levels of expertise. While popular first-person shooter games have been shown to improve cognitive abilities in young adults, Gazzaley says this happens by accident. NeuroRacer is a closed-loop game, in which the level of play is adjusted to the player’s behavior – and eventually to his or her own brain. The next version of the game, which Gazzaley is developing with Boston-based Akili Interactive Labs, where he is chief science adviser, will feature closed loops that adapt during every second of play. Gazzaley’s lab is also working on new games that employ transcranial electrical stimulation, a very mild shock targeted to particular parts of the brain to enhance learning. When playing one of these new games, the player receives low-frequency bursts of energy in certain parts of the frontal lobe. “We are studying if you learn faster if you play a game while we stimulate you at the right frequency,” Gazzaley explains.
The therapeutic and educational potential of such games is real and vast. They could be targeted, like NeuroRacer, to a healthy elderly population or be used as an educational tool in schools. Or they could be used to ameliorate known deficits in old and young alike. Gazzaley is currently working with pediatric neurologist Elysa Marco, PhD, on a game aimed at helping children with attention deficit disorder to better train their focus. The two are also teaming up to develop games for patients with autism, in an effort to stimulate the parts of their brains that the disorder has locked away.
New Blood
Gazzaley and Villeda come at cognition along very different paths, but with equally impressive vigor and results. And they are energized by each other’s work. “Adam’s games are incredible,” says Villeda. “Soon we will be able to grab an iPad and do games that will significantly improve our cognition. Who would have thought of that?”
Villeda sees parallels in their approaches to enhancing cognition during the aging process – through collaboration. He joined forces with bioinformaticians to help him sort through his data, with molecular biologists to create viruses, and with behavioral neuroscientists to identify the best ways to test cognition. “Immunology, neurobiology, and stem cell science all come together when talking about rejuvenation,” says Villeda. He believes that building bridges among disciplines will be critical for translating what is now fascinating research into the clinical realm.
“Saul’s and my research could be very synergistic in ways that we don’t fully understand right now, and Alvarez-Buylla’s work has been foundational to neuroscience,” says Gazzaley. “There is no Holy Grail for enhancing cognition, so what we probably should have been focusing on for the past 40 years is how the many interventions in our toolbox might interact with each other.”
Perhaps someday soon, baby boomers will be able to relive their 20s, at least cognitively, by taking a shot of Villeda’s revitalizing plasma while playing a video game developed by Gazzaley. Or maybe Alvarez-Buylla will have figured out how to engineer the perfect mix of neural stem cells to rebuild what age tears down. While we wait, Gazzaley urges us to apply all the strategies that science has already endorsed: Research has long shown that diet, exercise, and enriched, engaging environments are good for the brain. In fact, a new study out of the Cleveland Clinic showed that people with a genetic predisposition for Alzheimer’s were able to stave off neurologic decline with a three-day-a-week exercise routine. Those with the same disposition who chose not to get off the couch showed significant degeneration.
“Clearly the brain does not do well with comfort,” Gazzaley says, “so challenge it as much as you can.”