In a hospital room at UC San Francisco’s Parnassus campus, a patient anticipates having an epileptic seizure. A dozen wires trail from her gauze-wrapped head to a bedside monitoring device. Hidden beneath gauze and bone lies an intricate network of electrodes – each roughly the size of a flattened grain of rice. They’ve been surgically implanted on the surface of her brain, with the goal of revealing the origin of her complex, near-weekly seizures. At the same time, she is volunteering as a participant in groundbreaking research on mood disorders like depression, as well as related disorders like anxiety.
This patient is one of about 60 who receive similar specialized surgical treatment annually from UCSF neurosurgeon Edward Chang, MD ’04, because a cure for their epilepsy is unusually elusive. The electrodes stay in place for up to three weeks as Chang and his colleagues at the UCSF Epilepsy Center map which region of their brain triggers the seizures in the hope that they can be alleviated through surgery.
These implanted electrodes also present a rare opportunity to study an even more elusive target: the source of human moods and emotions.
Simply being alive can be excruciating for those who suffer from severe depression or anxiety. Many derive little enjoyment from family, work or play; some have a hard time functioning; and for a few, mood disorders are so irremediable that they take their own lives. Yet just as lasting happiness evades those with intractable mood and anxiety disorders, definitive treatments have evaded those hoping to help such patients. For some, drugs and other therapies fail to fully resolve their suffering. For a few, treatment fails entirely.
But what if such troubles stem from flawed brain circuitry, which could be coaxed to support healthy neural processing instead? A research project headed by Chang aims to answer this question – and change the way we understand and treat mood disorders.
If successful, the project will for the first time map the networks in the brain that have gone awry, then remodel them to restore healthy functioning using a new type of device that could be permanently implanted in the brain. When the pathways corresponding with depression are activated, the pattern of activity would be picked up by specialized electrodes. The electrodes, which are currently in development, would emit a minute impulse. The stimulation would encourage the brain to shift its activity into new, healthier patterns – possibly for good.
“This would be revolutionary,” says Vikram Rao, PhD ’06, MD ’08, an assistant professor of neurology who’s on Chang’s team. “The problem is not that the whole brain is malfunctioning; it’s certain key circuits that mediate mood. The device would be able to intervene on those circuits and restore them to a more healthy state.”
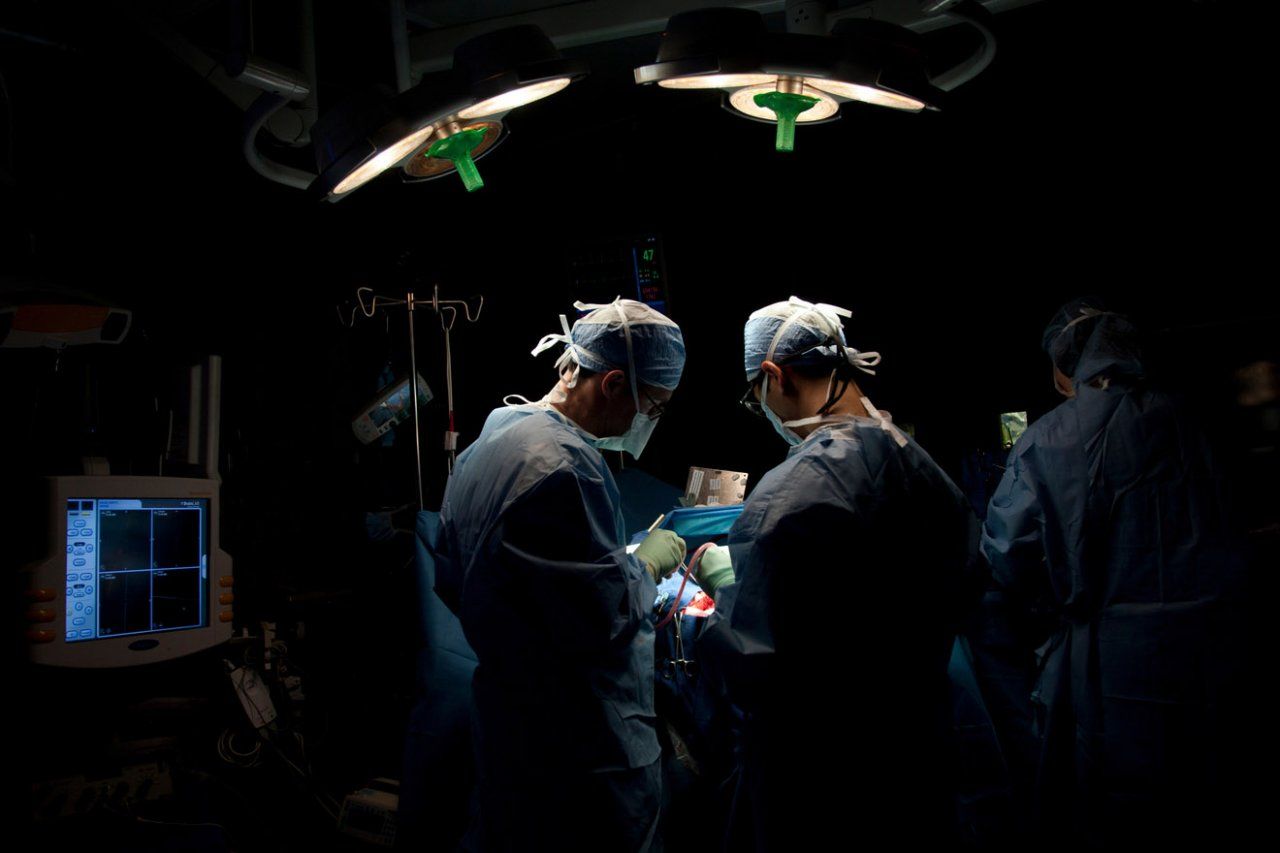
Neurosurgeon Eddie Chang (left) operates on a patient with epilepsy. Chang’s team is mapping the regions of the brain that trigger seizures while also studying the circuits that mediate mood. Photo: Cindy Chew
Collaborative Genesis
Chang is a lean, soft-spoken 39-year-old who first operated on a human brain in July 2004. From his early days as a medical student at UCSF, where he also completed his residency, he was attracted to the skill and compassion necessary for neurosurgery. By the time he started this project, he’d already made groundbreaking discoveries regarding how the brain processes language – using many of the same tools now being applied to understanding mood and anxiety disorders.
One October morning in 2013, he learned from the New York Times that the Defense Advanced Research Projects Agency (DARPA) planned to spend more than $70 million to advance research on brain implants and therapeutic brain stimulation – exactly his area of research.
Chang thought, “Okay, let’s get ready for this.”
“We had all heard about it, but Eddie is the one who said, ‘Do you want to do something about this?’” says Vikaas Sohal, MD, PhD, an assistant professor of psychiatry. “It’s definitely a team effort, but it’s his core vision of the themes and the people.”
That vision bore fruit; in the spring of 2014, the project was one of the first to be approved under President Obama’s Brain Initiative, which supports research seeking new ways to treat, prevent, and cure brain disorders. DARPA is a major funding partner of the Brain Initiative, known for sponsoring cutting-edge and high-risk projects. An initial $12 million in funding may go up to $26 million if the project meets key milestones.
“This is a huge privilege because we have the resources to do something that will have direct impact on patients – our project was chosen, but other good ones were not,” says Chang. “I think about that responsibility a lot, actually. Meeting this challenge can only be done at certain places in the world, and UCSF is one of them.”

Matthew State, chair of the UCSF Department of Psychiatry, says understanding the physical mechanisms of mental illness will help reduce its stigma. Photo: Stephan Babuljak
A project of this scope depends inherently on collaboration: a dozen labs in the fields of surgery, neurology, psychiatry, engineering and software development are all integral to its work. Contributing institutions include UCSF, UC Berkeley, the University of Southern California, Lawrence Livermore National Laboratory, and New York University, while Posit Science and Cortera Neurotechnologies are industry partners. Although academe is sometimes charged with a so-called “silo” mentality – an inclination to stay within departmental and disciplinary boundaries – this kind of cooperation among labs, specialties and institutions is a core value of the UCSF scientific community.
“One of the reasons I came to UCSF was that there was a tremendous opportunity to break down the silos and barriers between psychiatry, neurology and neurosurgery,” says Matthew State, MD, PhD, chair of the Department of Psychiatry and the Oberndorf Family Distinguished Professor. State was recruited from Yale in 2013 and since then has been actively fostering such collaborations to tackle challenges including autism, eating disorders, sleep and circadian rhythm disturbances, and interventional psychiatry – as well as mood disorders.
“I have a strong conviction that those [silos] have not served us well, and that we need to be thinking collectively about how to attack these problems and not be in our separate camps,” says State. His vision is spurring a groundswell of support, including philanthropy that’s putting mental health innovation on a new trajectory at UCSF.
When the Stars Align
The relationship between brain and mood has never before been looked at from such a detailed, structural standpoint. Doing so requires precisely siting electrodes on the surface of a living brain, slipping them under the skull like sliding a letter into an envelope.
“We are pioneering in this area in a way that is both ethical and a win-win for everyone involved,” Chang says.
We really want to go after this idea of using neural plasticity: remodeling the brain toward thinking about curative options.”
Patients who have come to UCSF for epilepsy treatment – as well as others whom UCSF neurosurgeon Philip Starr, MD, PhD, is treating for Parkinson’s – are key to the project. They’re having electrodes implanted out of medical necessity, but in parts of the brain that researchers want to examine because of their impact on mood. In addition, it happens that people with epilepsy and Parkinson’s also suffer from a much higher rate of intractable depression than the general population.
In other words, chance has given the team access to an ideal pool of research participants. All the scientists need is the patients’ consent to study their moods and emotions as they undergo treatment – consent that is often granted. “It’s remarkable and humbling to see the interest patients have in participating,” says Heather Dawes, PhD, project co-director. “They know it’s unlikely to benefit them directly, but they take the time to work with us anyway.”
The electrodes generate raw data that resembles an elaborate seismogram: multicolored lines that squiggle densely across the page, each representing activity in a different region of the brain.
That data is then correlated with patients’ moods: something inherently personal and subjective. Their feelings are quantified using an app that is based on research into how mood affects the performance of various concrete tasks. A tablet kept by patients’ bedsides pings four or five times a day, reminding them to run through a few electronic exercises designed for the project by the software firm Posit Science, which was co-founded by Michael Merzenich, PhD, an emeritus professor with whom Chang worked early in his UCSF training.
In addition to asking patients a series of self-reporting questions (such as whether they’re “depressed or happy” or “worthless or valuable”), the app also has them perform a variety of tasks. On one, depressed people exhibit slower reaction times. On another, they’re more apt to identify a face as sad rather than happy.
I feel we are on the brink of a transformative insight. . . . It could lead to totally different ways of treating depression.”
The results are then correlated with the real-time brain activity recorded by the electrodes. The goal is to develop a high-resolution map of which brain networks are activated in patients with mood disorders.
“I feel we are on the brink of a transformative insight,” Sohal says. “We are measuring things that honestly have scant examples of having been looked at in the context of depression. It could lead to totally different ways of treating depression from the way that we are right now.”

Vikaas Sohal (left) and Eddie Chang are uniting their fields of psychiatry and neuroscience to better understand mood and anxiety disorders. Photo: Stephan Babuljak
A Better Mousetrap
While the bulk of the data collection is taking place at UCSF, engineers at UC Berkeley, Cortera, and Lawrence Livermore are heading up efforts to develop specialized electrodes and deep-brain stimulation devices that can be safely implanted for long-term therapeutic use. To be successful, the device will have to be entirely self-contained within the brain – battery, electrodes, software and all – just as an artificial pacemaker is permanently situated within the chest.
“It needs to be biocompatible, lightweight, small, flexible, with good battery life and certain electrical characteristics,” Rao says. “That’s a real engineering feat.”
Furthermore, the array needs to be closed-loop, meaning it will record the brain’s activity, then selectively deliver an electrical stimulus based on abnormal rhythms it detects.
Even better would be if the device doesn’t need to be permanent. “Unlearning depression” is what Rao calls the possibility that targeted manipulation of key brain circuits could reset an individual’s mood to a more normal state. If the brain learns to use the new pathway without the electrical tweaks, the device could eventually be removed.
“We really want to go after this idea of using neural plasticity: remodeling the brain toward thinking about curative options,” Chang says.
It may be a long shot that all will go as planned. Yet after only one year, the team has already achieved some level of success – perhaps changing the landscape of mood research for good. “This treatment has a very promising therapeutic effect,” Chang says.
Chasing the Rainbow
When Chang speaks about mood disorders, he often opens his presentation with an image of the Golden Gate Bridge – and the observation that it is possibly the top suicide location in the U.S.
“It’s not getting better. In fact, 2013 was a record year for suicide attempts from the bridge,” Chang observes. “As far as we can tell, the burden of disease, and the suffering, have never been higher. ... Some even go for electro-convulsive therapy, where their whole brain is shocked. It turns out it’s very effective, but it’s only temporary and sometimes has negative effects on memories.”
The lay public tends to think of depression or anxiety as either a psychological weakness or a chemical imbalance. The latter view is due to highly publicized research on variations in neurotransmitters, such as serotonin and dopamine. In this view, the brain is simply awash in a sea of negative emotion; whole-body treatments such as drugs, therapy or exercise may, or may not, be able to turn that tide. But until recently, no one had been able to pinpoint the specific physical structures in the brain responsible for the problems; previous generations of neuroimaging tools simply weren’t up to the task. Arguably, the closest attempt was the pioneering work of Helen Mayberg, MD, a neurologist at Emory University who was able to achieve improvements in mood with deep-brain stimulation. However, she looked mostly at a single region of the brain, while the larger, network-level basis of mood disorders remains unclear. Furthermore, her approach was successful in small studies but thus far has failed in clinical trials, for unknown reasons.
“There are lots of different ways of conceptualizing depression, and we don’t know which one is right,” Sohal says. Yet refining the way we approach this issue isn’t an academic question for the millions of people worldwide who suffer from treatment-resistant psychiatric diseases.
“This particular collaboration is going to have an impact for an enormous number of our patients, starting with those suffering from depression but extending in the long run to those with anxiety, psychosis and other such disorders,” State says.
If it’s successful, the project is likely to radically change the way depression is diagnosed and treated. Rather than relying on verbal descriptions of symptoms, diagnoses would be based on identifying specific maladapted circuits as the cause of various crippling mood disorders. Treatment could then be targeted accordingly. Overall, mood could come to be seen as a physical, not a moral failing: more akin to a broken bone than a broken heart. If such a paradigm shift occurs, Chang and his team foresee a secondary benefit for all patients with mood disorders, large or small: reduced stigma.
“Having knowledge about the physical mechanism of depression will help everybody view it differently,” Sohal says, adding that many of his friends, family members and even patients currently feel that depression is not something real.
“As we make progress in this area, the stigma and marginalization that are just part and parcel of having a psychiatric illness will start to fade, I guarantee you,” State says. “It is going to have an enormous impact.”